A Year of Reckoning for Novo Nordisk
The Danish pharmaceutical giant, Novo Nordisk, finds itself at a critical juncture as it navigates the turbulent waters of 2026. Once a market darling, the company has recently experienced a significant downturn, marked by a volatile share price and a challenging year on record. With patent expiries looming in key markets like Brazil, Canada, and China, coupled with mounting pricing pressures, the stage is set for a transitional period that analysts deem a “show me case” for investors, particularly in the crucial U.S. market.
Navigating Turbulent Waters
Novo Nordisk’s recent struggles stem from a confluence of factors: a series of downward guidance revisions, the aggressive strides of chief rival Eli Lilly, a leadership shake-up, and the proliferation of more affordable generic alternatives impacting the vital U.S. market. These headwinds have eroded investor confidence, making the upcoming year a defining moment for the company’s future trajectory in the lucrative weight loss sector.
The Oral Revolution: Wegovy’s Game-Changing Approval
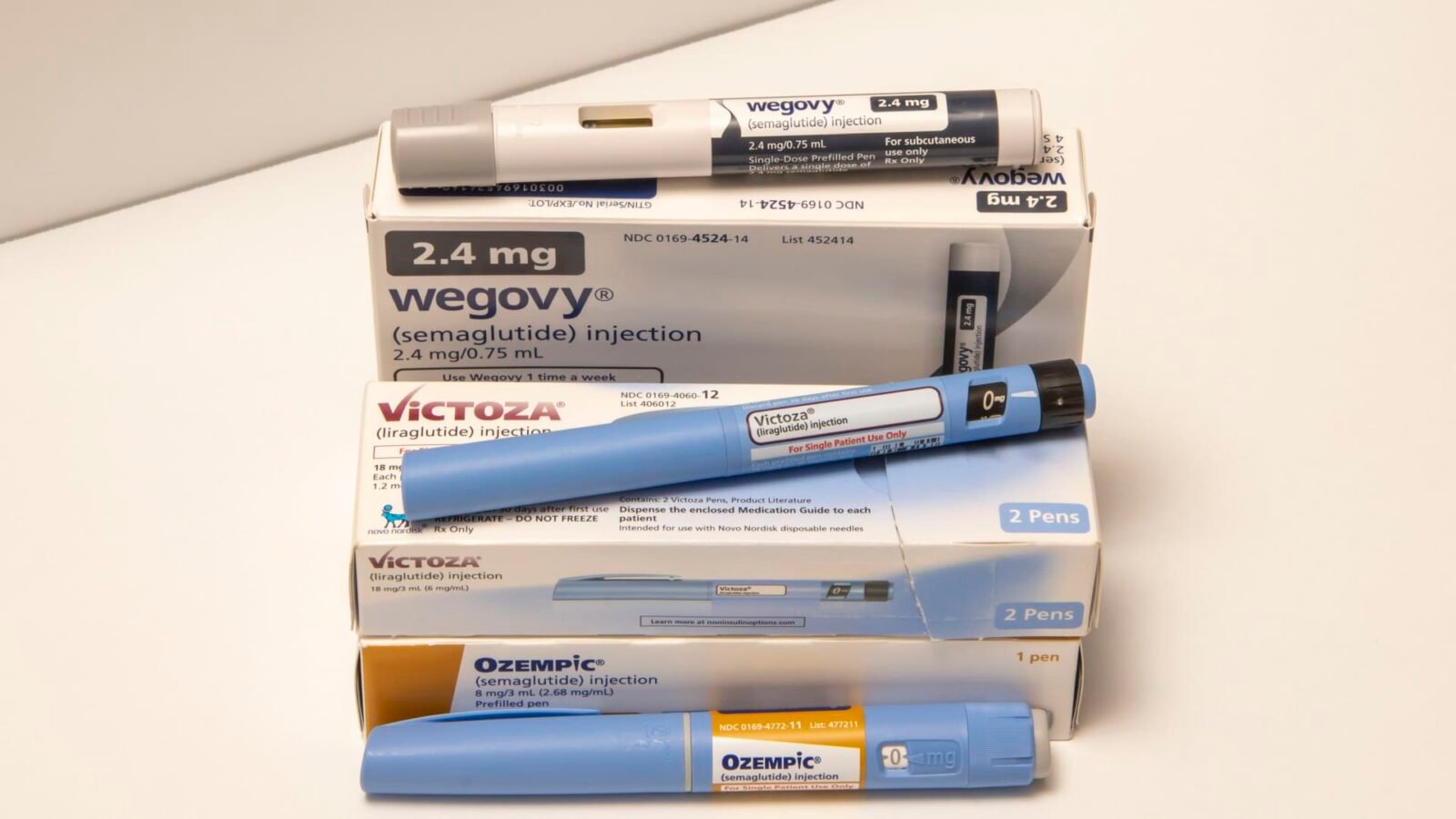
Just as 2026 dawned, Novo Nordisk delivered a strategic counterpunch with the U.S. approval of its new oral weight loss pill, branded Wegovy. This landmark decision marks the first oral GLP-1 treatment sanctioned for weight loss, sending Novo’s shares soaring by nearly 10%. Investors are banking on this innovation to help the company reclaim lost ground and fend off fierce competition from Eli Lilly and other contenders.
This “early Christmas present,” as one analyst aptly described it, underscores the pivotal themes that will shape Novo Nordisk’s performance this year: a strategic shift from injectables to oral medications and the intense race for market dominance in the GLP-1 space.
Pill Power: Efficacy Meets Convenience
The introduction of an oral option could be a significant differentiator for Novo Nordisk. While the approval was largely anticipated, its impact is undeniable. The oral version of Wegovy has demonstrated impressive efficacy, with patients achieving an average body weight reduction of 16.6% over 64 weeks. This stands favorably against Eli Lilly’s forthcoming orforglipron pill, which averages 12.4% over 72 weeks.
Crucially, Novo Nordisk CEO Mike Doustdar highlighted a rare synergy: “Wegovy in a pill basically will have the same efficacy as its injectable counterpart.” This combination of high efficacy with the inherent convenience of a pill—no need for cold storage, simpler distribution, and easier market entry—is expected to resonate strongly with consumers, potentially allowing Novo Nordisk to recapture market share and drive growth.
The Looming Battle with Eli Lilly
The competitive landscape remains intense. Eli Lilly is poised to receive FDA approval for its own weight loss pill, orforglipron, by the second quarter of this year. The ensuing head-to-head competition will be closely watched by investors, as both pharmaceutical giants vie for supremacy in the rapidly expanding oral GLP-1 market.
Redefining the Narrative: Beyond Weight Loss
Eli Lilly has successfully positioned its rival injectable drug, Zepbound, as the market leader for maximum weight loss. In contrast, Novo Nordisk has often emphasized a broader narrative, advocating for obesity to be recognized as a chronic disease and highlighting Wegovy’s positive impact on related comorbidities.
Market Demands vs. Medical Philosophy
Novo’s strategy focuses on developing assets that address multiple health issues, as Doustdar noted: “We have seen that with semaglutide; it helps liver, kidney, heart – that’s fantastic – we should actually go and further develop those.” However, market sentiment, particularly in the U.S., appears to prioritize significant weight loss. Sydbank analyst Søren Løntoft Hansen suggests that the fraction of prescriptions for obesity drugs tied to obesity-related diseases is minimal, and patients, even if not aiming for extreme weight loss, desire the *option* for the highest possible reduction.
This disparity between medical philosophy and market demand presents a challenge. If Novo Nordisk can effectively integrate the appeal of high weight loss efficacy with its broader disease management narrative, especially with the convenience of the Wegovy pill, it could be well-positioned to regain its footing in this fiercely competitive arena.
The Path Forward: A Must-Win Scenario
For Novo Nordisk, 2026 is undeniably a year of defense, but also one of immense opportunity. The success of the oral Wegovy in the U.S. market is not merely about product launch; it’s about restoring investor confidence, fending off a formidable rival, and solidifying its position in the future of obesity treatment. The stakes are high, and the battle for market leadership is a must-win for the Danish drugmaker.
For more details, visit our website.
Source: Link